Press Release
Amnovis expands to the United States and acquires Westconn Precision Technologies’ additive manufacturing activities
- Chris Cook appointed General Manager, US Operations for Amnovis’ new production site in the Warsaw, Indiana area
-
Acquisition of additive manufacturing activities of Westconn Precision Technologies brings Jake Marasco to Amnovis, strengthening North American commercial presence and hybrid manufacturing expertise
- Customers will benefit from globally deployable manufacturing capacity enabled by mirrored US and European operations, significantly simplifying supplier qualification requirements
Aarschot, Belgium - April 29, 2026
Amnovis, a Belgian additive manufacturing contract manufacturing organization serving regulated industries including medical devices, announces the establishment of a new operational entity in the United States and the acquisition of the additive manufacturing activities of Westconn Precision Technologies. Together, these steps represent a key milestone in Amnovis’ strategy to build a global additive manufacturing organization with harmonized operations across Europe and North America.
The new US operations will be located in North Webster, Indiana, and directly within the unique ecosystem surrounding Warsaw, Indiana. The site is being developed as a mirror site of Amnovis’ Belgian headquarters, operating under the same global quality management system, procedures, digital workflows, validation frameworks and equipment strategy.

To lead the US operations, Amnovis has appointed Chris Cook as General Manager, US Operations. Cook brings extensive leadership experience across scale ups and large international medical device organizations, and will oversee the ramp up and operational execution of Amnovis’ US manufacturing activities.
“Expanding our operational footprint to the United States is a logical next step in the evolution of Amnovis,” said Ruben Wauthle, CEO and Co founder of Amnovis. “Being close to the largest medical device market globally, and specifically within the Warsaw, Indiana ecosystem, allows us to better support customers who require reliable, high quality additive manufacturing capacity with minimal logistical complexity.”
By establishing mirrored production sites in Europe and the United States under a single global quality management system, Amnovis enables customers to deploy manufacturing capacity across regions while maintaining consistent processes, equipment and validation approaches. This operating model is designed to support flexible and scalable manufacturing in regulated environments.

As part of its US expansion, Amnovis has acquired the additive manufacturing activities of Westconn Precision Technologies. These activities will be integrated into Amnovis’ new US operations in North Webster, Indiana, strengthening its North American footprint and hybrid manufacturing capabilities by combining metal additive manufacturing with conventional CNC and post processing technologies.
In support of this expansion, Jake Marasco will join Amnovis and take responsibility for account management and business development in North America. Marasco brings deep experience in integrating additive manufacturing into broader manufacturing workflows, with a strong focus on combining additive manufacturing and precision machining strategies.
“The acquisition of Westconn’s additive manufacturing activities further strengthens Amnovis’ position as a global partner for industrialized additive manufacturing,” said Ruben Wauthle. “By aligning equipment strategies and technical expertise across both sites, we are able to scale our operating model globally while further reinforcing our already strong capabilities in metal additive manufacturing. This combination allows us to support customers with a level of consistency, scale and technical depth that is increasingly required as more customers develop applications that truly unlock the full potential of additive manufacturing.”
Marasco adds, “Westconn’s additive manufacturing work has been built around helping customers move complex programs from development into reliable production. Joining Amnovis creates a strong path to continue supporting those customers with deeper additive manufacturing expertise, a broader global platform, and a clear commitment to building U.S.-based capacity.”
The US expansion is driven by long term market dynamics, including the size of the US medical device market, the growing demand for patient specific and time critical applications, and the need for flexible, regionally available manufacturing capacity. With operations now spanning Europe and the United States, Amnovis continues to execute on its ambition to become a leading global additive manufacturing contract manufacturing organization for regulated and high tech industries.
ABOUT AMNOVIS
Amnovis is an independent, innovation-driven additive manufacturing company specializing in high-end 3D-printed products for regulated and high-tech industries, with a strong focus on medical devices. Leveraging deep process and material expertise, Amnovis delivers state-of-the-art solutions, from patient-specific implants to scalable volume production. With a fully digital, ISO 13485:2016-certified workflow, Amnovis partners with OEMs worldwide to accelerate product development, streamline validation, and raise the bar in additive manufacturing. By bridging emerging technologies with real-world applications, Amnovis continuously redefines the possibilities of 3D printing.
For more information, visit www.amnovis.com or follow Amnovis on LinkedIn.
MEDIA CONTACT
Ruben Wauthle, CEO & Co-founder - info@amnovis.com - +32 16 14 60 76
MEDIA PACKAGE
Media package with pictures optimized for online use can be downloaded from the link below. For high resolution pictures, please contact us.
Amnovis logo to be used for white backgrounds:

Amnovis logo to be used for other backgrounds:

Amnovis PR-260429 Figure 1: Chris Cook, General Manager, US Operations, in front of the Amnovis facility in North Webster, Indiana:

Amnovis PR-260429 Figure 2: Selected L-PBF metal 3D printers installed at Amnovis:

Amnovis PR-260429 Figure 3-4: Additive manufacturing of medical devices in action:


Amnovis PR-260429 Figure 5: Titanium spinal implants immediately after printing:

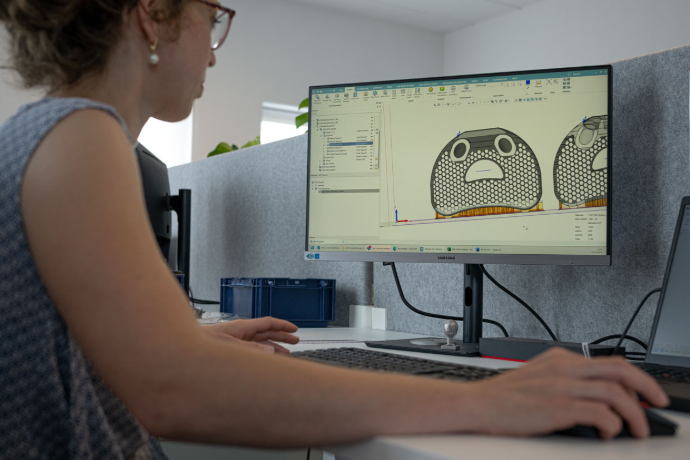
Amnovis PR-260429 Figure 6: An application engineer preparing files for metal additive manufacturing: